Main Article Content
Abstract
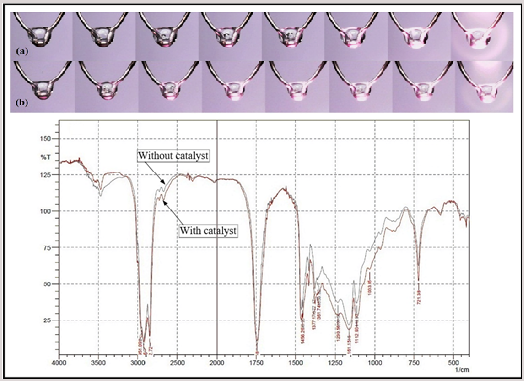
This study examines the Ignition characteristics of blended fuel droplets with crude coconut oil and rhodium liquid as a liquid metal catalyst. The ignition behavior was observed by igniting the oil droplet on a junction of a thermocouple, and the droplet evolution recorded with the high-speed camera. The results show that the addition of a liquid metal catalyst successfully reduces the molecular mass of the triglyceride and weakens the bonding force between the carbon chain, and therefore the viscosity and flash point decreases. Moreover, the addition of liquid metal catalysts increased the reactivity of fuel molecules such as C-H, C-C, C = C, and C-O. Changes in the physical properties of the fuel, the geometry of the carbon chain, and molecular mass ease the absorption of heat by the fuel droplet, thereby increasing fuel ignition performances.
Keywords
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
- S. Che Mat, M. Y. Idroas, M. F. Hamid, and Z. A. Zainal, “Performance and emissions of straight vegetable oils and its blends as a fuel in diesel engine: A review,” Renewable and Sustainable Energy Reviews, vol. 82, pp. 808–823, 2018.
- IEA, “World Energy Outlook 2019,” 2019. [Online]. Available: https://www.iea.org/reports/world-energy-outlook-2019. [Accessed: 05-May-2020].
- L. F. Bautista, G. Vicente, R. Rodríguez, and M. Pacheco, “Optimisation of FAME production from waste cooking oil for biodiesel use,” Biomass and Bioenergy, vol. 33, no. 5, pp. 862–872, 2009.
- D. Ayu, R. Aulyana, E. W. Astuti, K. Kusmiyati, and N. Hidayati, “Catalytic Transesterification of Used Cooking Oil to Biodiesel: Effect of Oil-Methanol Molar Ratio and Reaction Time,” Automotive Experiences, vol. 2, no. 3, pp. 73–77, 2019.
- A. C. Arifin, A. Aminudin, and R. M. Putra, “Diesel-Biodiesel Blend on Engine Performance: An Experimental Study,” Automotive Experiences, vol. 2, no. 3, pp. 91–96, 2019.
- E. Marlina, M. Basjir, M. Ichiyanagi, T. Suzuki, G. J. Gotama, and W. Anggono, “The Role of Eucalyptus Oil in Crude Palm Oil As Biodiesel Fuel,” Automotive Experiences, vol. 3, no. 1, pp. 33–38, 2020.
- C. K. Law, “Recent advances in droplet vaporization and combustion,” Progress in Energy and Combustion Science, vol. 8, no. 3, pp. 171–201, 1982.
- H. F. Ramalho et al., “Biphasic hydroformylation of soybean biodiesel using a rhodium complex dissolved in ionic liquid,” Industrial Crops and Products, vol. 52, pp. 211–218, 2014.
- C. Vangelis, A. Bouriazos, S. Sotiriou, M. Samorski, B. Gutsche, and G. Papadogianakis, “Catalytic conversions in green aqueous media: Highly efficient biphasic hydrogenation of benzene to cyclohexane catalyzed by Rh/TPPTS complexes,” Journal of Catalysis, vol. 274, no. 1, pp. 21–28, 2010.
- A. Bouriazos, S. Sotiriou, C. Vangelis, and G. Papadogianakis, “Catalytic conversions in green aqueous media: Part 4. Selective hydrogenation of polyunsaturated methyl esters of vegetable oils for upgrading biodiesel,” Journal of Organometallic Chemistry, vol. 695, no. 3, pp. 327–337, 2010.
- M. Zhu, Y. Ma, and D. Zhang, “Effect of a homogeneous combustion catalyst on combustion characteristics of single droplets of diesel and biodiesel,” Proceedings of the Combustion Institute, vol. 34, no. 1, pp. 1537–1544, 2013.
- Z. Ma, Y. Li, Z. Li, W. Du, Z. Yin, and S. Xu, “Evaporation and combustion characteristics of hydrocarbon fuel droplet in sub- and super-critical environments,” Fuel, vol. 220, pp. 763–768, 2018.
- A. M.-D. Faik and Y. Zhang, “Multicomponent fuel droplet combustion investigation using magnified high speed backlighting and shadowgraph imaging,” Fuel, vol. 221, pp. 89–109, 2018.
- M. Al Qubeissi, S. S. Sazhin, and A. E. Elwardany, “Modelling of blended Diesel and biodiesel fuel droplet heating and evaporation,” Fuel, vol. 187, pp. 349–355, 2017.
- Y. Xu, I. Keresztes, A. M. Condo, D. Phillips, P. Pepiot, and C. T. Avedisian, “Droplet combustion characteristics of algae-derived renewable diesel, conventional #2 diesel, and their mixtures,” Fuel, vol. 167, pp. 295–305, 2016.
- F. K. Crundwell, M. S. Moats, V. Ramachandran, T. G. Robinson, and W. G. Davenport, “Chapter 31 - Platinum-Group Metals, Production, Use and Extraction Costs,” Oxford: Elsevier, 2011, pp. 395–409.
- I. Iavicoli and V. Leso, “Rhodium,” in Handbook on the Toxicology of Metals, Elsevier, 2015, pp. 1143–1174.
- M. Shelef and G. W. Graham, “Why rhodium in automotive three-way catalysts?,” Catalysis Reviews, vol. 36, no. 3, pp. 433–457, 1994.
- J. P. Loferski, “Commodity Report: Platinum-Group Metals, United States Geological Survey. Retrieved July 16, 2012.(b) DA Colby, RG Bergman and JA Ellman, 2010,” Chem Rev, vol. 110, p. 624, 2013.
- K. Ashley and R. Fairfax, “Sampling and analysis of soluble metal compounds,” NIOSH Manual of Analytical Methods, vol. 4, pp. 167–178, 2003.
- Q. Zhou et al., “Nano La2O3 as a heterogeneous catalyst for biodiesel synthesis by transesterification of Jatropha curcas L. oil,” Journal of Industrial and Engineering Chemistry, vol. 31, pp. 385–392, 2015.
- H. Y. Nanlohy, I. N. G. Wardana, N. Hamidi, L. Yuliati, and T. Ueda, “The effect of Rh3+ catalyst on the combustion characteristics of crude vegetable oil droplets,” Fuel, vol. 220, pp. 220–232, 2018.
- H. Y. Nanlohy, I. N. G. Wardana, N. Hamidi, and L. Yuliati, “Combustion characteristics of crude jatropha oil droplets using rhodium liquid as a homogeneous combustion catalyst,” in IOP Conference Series: Materials Science and Engineering, 2018, vol. 299, no. 1, p. 12090.
- National Center for Biotechnology Information, “Rhodium(II) sulfate CID=14252067,” PubChem Database. [Online]. Available: https://pubchem.ncbi.nlm.nih.gov/compound/Rhodium_II_-sulfate. [Accessed: 05-May-2020].
- M. Mehl, W. J. Pitz, C. K. Westbrook, K. Yasunaga, C. Conroy, and H. J. Curran, “Autoignition behavior of unsaturated hydrocarbons in the low and high temperature regions,” Proceedings of the Combustion Institute, vol. 33, no. 1, pp. 201–208, 2011.
- A. Hoxie, R. Schoo, and J. Braden, “Microexplosive combustion behavior of blended soybean oil and butanol droplets,” Fuel, vol. 120, pp. 22–29, 2014.
- C. V Naik, C. K. Westbrook, O. Herbinet, W. J. Pitz, and M. Mehl, “Detailed chemical kinetic reaction mechanism for biodiesel components methyl stearate and methyl oleate,” Proceedings of the Combustion Institute, vol. 33, no. 1, pp. 383–389, 2011.
- C. K. Westbrook et al., “Detailed chemical kinetic reaction mechanisms for soy and rapeseed biodiesel fuels,” Combustion and Flame, vol. 158, no. 4, pp. 742–755, 2011.